DEFRA have released guidance about the ‘oval’ health marks applied to products of animal origin (POAO) for approved establishments.
Currently, those premises approved by the Food Standards Agency (FSA) and subject to EU regulation 853/2004 are required to place a health mark on their products. Companies under the control of the environmental health department of their local council (eg those not handling raw materials) may be exempt from this requirement.
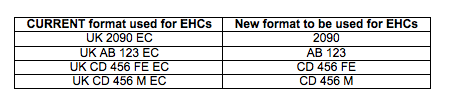
The current format for a health mark is: – UK xxxx EC (where the xxxx is either 4 numerals or 2 letters and 3 numerals).

The following changes will be implemented whatever the outcome of Brexit.
For MEAT products to AUSTRALIA, CANADA, JAPAN and USA – these changes will come into effect on 31st August 2019
For –
MEAT products to SOUTH KOREA, SAUDI ARABIA, SINGAPORE, SOUTH AFRICA, TAIWAN and THAILAND;
DAIRY products to BRAZIL, CHILE, INDONESIA, MALAYSIA;
FISH products to BRAZIL – these changes will come into effect on 30th September 2019
For PORK, DAIRY, FISH products to CHINA – date of change is still to be confirmed.
ALL other products/commodities the changes will occur from the date the UK leaves the EU, currently due to be 31st October 2019
From the dates specified, any reference to the approval number on the EHC must reflect the new format. Current APHA guidance is that the goods themselves do NOT require relabelling after these dates and companies should continue to follow FSA guidance on this matter.
We further understand there is a 6 month crossover period where marks in either format will be accepted, but it would be good practice to start using the new health marks from the dates stipulated.
Exporters should update any templates for EHC applications with the new health marks at the appropriate times.
Should you have any further queries please do not hesitate to contact us.
Written by Andrew Iveson on and tagged in health mark, .